Posted: July 25, 2024 | Author: Karin Verzijden | Filed under: Advertising, alternative protein, Authors, Enforcement, Food, Information |
 On 23 July 2024 a Dutch Court ruled in summary relief proceedings that Upfield cannot use the name “roombeter” for a plant-based alternative for butter, as this is in violation of the Regulation establishing a common organization of the markets for agricultural products (“COM Regulation”). You should know that “roombeter” translates in English as a composition of “cream” and “better”, whereas “cream” is a reserved designation under the COM Regulation that can only be used for dairy products. Furthermore, “beter” is close to “boter”, being the Dutch designation for butter.
On 23 July 2024 a Dutch Court ruled in summary relief proceedings that Upfield cannot use the name “roombeter” for a plant-based alternative for butter, as this is in violation of the Regulation establishing a common organization of the markets for agricultural products (“COM Regulation”). You should know that “roombeter” translates in English as a composition of “cream” and “better”, whereas “cream” is a reserved designation under the COM Regulation that can only be used for dairy products. Furthermore, “beter” is close to “boter”, being the Dutch designation for butter.
Facts of the case at hand
In the case at hand, Upfield markets a plant-based alternative for butter under the brand BLUE BAND and the product name ROOMBETER. The packaging of the product furthermore states “100 % plant-based alternative for butter” and “81 % less climate impact than butter”. The packaging itself consists of golden coloured paper that is also used for conventional butter in the Netherlands and it displays a curl of butter as shown below. The Dutch Dairy Association opposed the use of the product name ROOMBETER, as it is considered this a violation of the COM Regulation, as explained below.
Case before Dutch Advertisement Code Committee
Prior to this legal procedure , the Dutch Dairy Association had submitted a complaint regarding this product before the Dutch Advertisement Code Committee. This self-regulatory body ruled on March 21 last that the presentation of the product was misleading, since it could be understood to contain butter. The “e” in “beter” could be confused for an “o”, resulting in “boter”, which is Dutch for butter. And furthermore the golden coloured packaging added to the misleading character of the product. The topic of violation of the COM Regulation was left to civil law proceedings, as it exceeded the competence of the Committee.
Applicable legislation
Article 78.2 of the COM Regulation states that the definitions, designations and sales descriptions provided for in its Annex VII may be used in the Union only for the marketing of a product that conforms to the corresponding requirements laid down in that Annex. Annex VII contains, amongst other things, a product definition for milk and a list of milk products. It furthermore states that these designations may not be used for any other product than milk and milk products. The purpose of this provision is to protect dairy names from being used for non-dairy products.
Tofutown
You may recall that in its Tofutown decision back in 2017, the ECJ formulated a very strict prohibition of the use of diary names for non-dairy products (check out our blog on this case here). As a result of that prohibition, the use of the designation “Tofubutter” for a tofu-based product was in violation of the COM Regulation. As a general rule, the ECJ precluded the term ‘milk’ and the designations reserved by the COM Regulation exclusively for milk products from being used to designate a purely plant based product in marketing or advertising. This even applies if those terms are expanded upon by clarifying or descriptive terms indicating the plant origin of the product at issue
Arguments in favour of ROOMBETER
Upfield had argued it did not market its product under the designation ROOMBETER but under the designation BLUE BAND ROOMBETER. The brand BLUE BAND has been used for more than 100 years for margarine, so it is obvious for the consumer this is a plant-based product This is even strengthened by the Dutch translation of the designations “100 % plant-based alternative for butter” and “81 % less climate impact than butter”. So the name ROOMBETER does not designate, imply or suggest it is about a dairy product.
Court decision
The Court did not eat it. Instead, it very strictly applied the Tofutown doctrine, stating that a reserved designation under the COM Regulation cannot be used for a plant-based product. It went on to explain that if it is prohibited to use the designation “tofubutter” for a plant-based product, as it contains the reserved designation “butter”, for sure it is prohibited to use the designation “roombeter” for a plant-based product, as it contains another reserved designation under the COM Regulation. Also, the element “beter” (“better” in English) can hardly be perceived as a clarifying or descriptive term, as it does not refer (contrary to “tofu”) to a plant-based origin. In fact, its reference to plant-based origins can only be understood by those consumers who know the “skip the cow” ad or who further study the packaging of this product.
Upfield was therefore ordered to stop using the designation ROOMBETER within three months after the date of the legal decision.
Consequences of this decision
Should the conclusion of this decision be that any reference to dairy products should be meticulously avoided when marketing plant-based dairy replacements? This seems a very hard task, as manufacturers of these replacement products will want to indicate how their products can be used. Happily, this is not the case. It is still permitted to mention that your plant-based product is for instance a “yoghurt variation”, as this is perceived as a product explanation rather than a product designation. This is not in violation of the Tofutown doctrine and in line with a 2019 Dutch Supreme Court decision relating to a soy-based product marketed by Alpro. Advertising plant-based dairy alternatives nevertheless remains a delicate balancing act.
Posted: July 23, 2024 | Author: Jasmin Buijs | Filed under: Advertising, alternative protein, Food, Information |
 Claims for formula milk that refer to nature can be understood as a (prohibited) discouragement of breastfeeding, as was recently ruled in two instances by the Dutch Advertising Code Foundation. This case captured our attention, especially with an eye to future possibilities of cell-based breastmilk alternatives.
Claims for formula milk that refer to nature can be understood as a (prohibited) discouragement of breastfeeding, as was recently ruled in two instances by the Dutch Advertising Code Foundation. This case captured our attention, especially with an eye to future possibilities of cell-based breastmilk alternatives.
What is the case about? Advertiser, a food business marketing infant and follow-on formula, published statements on its website about its products such as:
- “Unique combination of natural lactic acid cultures and valuable fibers.”
- “Where organic ends, […] continues: […] guarantees an excellent quality and supersedes the legal requirements concerning organic.”
- “Inspired by nature.”
- “With the first milk that is non mom’s, I want to do all well” (next to a picture of a baby in the arms of a woman).
The question is whether (a) these texts qualify as advertisements, and (b) whether the texts give the impression that the follow-on formula is as good as breastmilk.
What is the legal rule? Consumer-oriented advertising for infant formula is prohibited. Advertisements for follow-on formula are possible, but should not dissuade or discourage breastfeeding. This is laid down in articles 4.1 and 5.2 of the Dutch Advertising Code for Infant Formula, in line with EU Regulation 2016/127. As follows from article 6(6) of aforementioned Regulation, the labelling, presentation and advertising of infant and follow-on formula shall not include terms like ‘humanized’, ‘maternalized’ or ‘adapted’. The Dutch Advertising Code for Infant Formula further lists ‘inspired by breastmilk’, ‘protected effect of breastmilk’, and ‘contains nutrients that are also found in breastmilk’ as examples of prohibited statements to avoid discouragement of breastfeeding.
What was decided? The Board of Appeal upheld the decision of the Advertising Code Committee and ruled that (a) the texts on advertiser’s website are clearly promotional in nature, and that (b) the consumer will understand the references to nature to be references to breastmilk (and not to the products having organic qualities, as was explained by advertiser). As this is in violation of the Dutch Advertising Code for Infant Formula, advertiser is requested to no longer make such advertisements.
 Our analysis and future outlook: It is commonly agreed that breastfeeding, where possible, should be supported. This is one of the principles laid down in the WHO International Code of Marketing of Breast-Milk Substitutes and the subsequent relevant resolutions of the World Health Assembly. We are therefore not surprised by the ruling.
Our analysis and future outlook: It is commonly agreed that breastfeeding, where possible, should be supported. This is one of the principles laid down in the WHO International Code of Marketing of Breast-Milk Substitutes and the subsequent relevant resolutions of the World Health Assembly. We are therefore not surprised by the ruling.
Looking into the future, it is interesting to note that the same rules shall in principle apply to cultivated breastmilk made in a lab. Various companies are already working on this concept such as French Nūmi and BIOMILQ in the US. Giants like Danone and FrieslandCampina have announced strategic partnerships with cell-based human milk component start-ups. Companies working on such substitute products obviously want to (and should) explain their products to the public. Statements such as ‘inspired by breastmilk’ and ‘contains nutrients that are also found in breastmilk’ may not seem unreasonable in this context. Currently this is however not allowed. Will the Dutch self-regulatory code (which is more specific in prohibited terms than EU Regulation 2016/127 and the WHO Code) be updated, so that such innovations can be appropriately explained to the public? Stay tuned – we will keep you posted!
The full case can be read here (in Dutch).
Posted: April 24, 2024 | Author: Jasmin Buijs | Filed under: Advertising, Authors, Food, Food Supplements, Health claims, Information, Nutrition claims |
 The International Probiotics Association Europe (IPA Europe) is calling for harmonized use of the claim ‘probiotics’ in the EU. Aforementioned term is generally considered an unauthorized health claim under the EU Claims Regulation. Nevertheless, an increasing number of EU member states allows the use of this claim under certain conditions. This blogpost dives into the regulatory status of probiotic claims in different EU member states and the latest developments in this area.
The International Probiotics Association Europe (IPA Europe) is calling for harmonized use of the claim ‘probiotics’ in the EU. Aforementioned term is generally considered an unauthorized health claim under the EU Claims Regulation. Nevertheless, an increasing number of EU member states allows the use of this claim under certain conditions. This blogpost dives into the regulatory status of probiotic claims in different EU member states and the latest developments in this area.
When entering the keyword ‘probiotic’ in the EU Health Claims Register, one is confronted with over 100 rejected health claims. Over the past years, this has led to the question whether the term ‘probiotic’ should be allowed under certain conditions, and has resulted into divergent policies in different EU member states. To protect both the food industry (against unfair competition) and the consumer (against misleading information), these divergent policies are reason for IPA Europe to call for a harmonized framework.
Czech Republic, Northern Ireland and France
Although EU member states generally consider ‘probiotics’ to be an unauthorized health claim under the EU Claims Regulation, some EU member states take a different approach. ‘Probiotics’ is for example considered a nutrition claim in the Czech Republic. Such claim is allowed if the conditions set forth in the EU Claims Regulation are met. This means, amongst others, that the good bacteria in question are present in the food in an amount that, according to generally accepted scientific evidence, causes the claimed beneficial effect. By contrast, in France and Northern Ireland, the term ‘probiotics’ can be used as a general, non-specific health claim that is allowed in combination with the authorized health claim “live cultures in yoghurt or fermented milk improve lactose digestion of the product in individuals who have difficulty digesting lactose”.
Italy
Italy takes the view that the term ‘probiotics’ does not meet the definition of a health claim and therefore falls outside the scope of the EU Claims Regulation. Italy supports this view by EFSA’s conclusion that probiotic colonization in the intestinal flora (without further specification of bacterial species or strains) is insufficient evidence to substantiate a beneficial effect on human health. In other words, no link between probiotics and health can be demonstrated, for which reason ‘probiotics’ cannot be considered a health claim. Aforementioned reasoning is however not a free pass to unconditionally use the term ‘probiotics’ in Italy. Instead, the following conditions must be met if and when using this term: (i) safety for human consumption, (ii) a history of use for the benefit of the intestinal flora, and (iii) presence of the relevant bacteria in the food in live form and in an adequate quantity until end of shelf life.
Denmark and Spain
In Denmark, the term ‘probiotics’ can be used based on a different legal ground, namely as a mandatory category designation under the EU Food Supplements Directive. In France, such is also possible. Indeed, Article 6(3)(a) of the EU Food Supplements Directive requires that the labeling of the food supplement shall bear “the names of the categories of nutrients or substances that characterize the product or an indication of the nature of those nutrients or substances”. In Spain, the claim ‘probiotics’ is allowed thanks to the principle of mutual recognition. Based on this principle, a product lawfully marketed in one EU member state must also be accepted on the market of another EU member state. Since probiotic claims are therefore allowed on the Spanish market for products from other EU member states, banning the term ‘probiotics’ at national level would discriminate against national producers. Therefore, both food products produced within Spain and those produced outside of the country can bear the claim ‘probiotics’.
Netherlands
In the Netherlands – just like in Denmark and France – the term ‘probiotics’ can be used as a category designation for food supplements. This Dutch practice is laid down in the Guideline Document on the EU Claims Regulation by the Dutch Health Advertising Knowledge and Advice Council (in Dutch: Keuringsraad). An earlier version of the Nutrition and Health Claims Manual by the Dutch Food Safety Authority (in Dutch: NVWA Handboek Voedings- en Gezondheidsclaims) also explicitly mentioned this possibility. Such is now longer mentioned in the latest version of the Nutrition and Health Claims Manual, which can be explained by the fact that ‘probiotics’ as a category designation for food supplements is not a nutrition or health claim.
Although claims that further elaborate on the health effect of probiotics (sporadically) occur on the Dutch (online) market, these are not allowed. Whether the expression “increases the good bacteria in the intestinal flora” (without using the term ‘probiotics’) is acceptable in the Netherlands, is yet unclear. Yakult uses this expression to advertise its fermented milk drink. The Dutch Health Advertising Knowledge and Advice Council would not allow this expression in the context of its preventive supervision in the context of food supplements, because this expression in effect creates a link between the food product and health. If the NVWA has however a different opinion on this and does consider aforementioned expression possible, then other food businesses can take advantage of this too. The call for harmonized uses of the term ‘probiotics’ by IPA Europe, about which more below, can possibly contribute to the acceptance of such expression at national level.
”Probiotics’ not a health claim”
As demonstrated above, various EU member states have introduced national rules on the use of the term ‘probiotics’. As a result, IPA Europe believes that the European Commission’s position that ‘probiotics’ implies a health benefit and is therefore a(n unauthorized) health claim no longer holds water. Instead, IPA Europe advocates qualifying the claim ‘contains probiotics’ as a nutrition claim, just like ‘contains vitamins and minerals’ and ‘contains fiber’. Aforementioned substances may have beneficial nutritional properties, but no specific health benefit is claimed. To strengthen its argument that ‘probiotics’ is not a health claim, IPA Europe explains that the term is not sufficiently precise to substantiate the claimed health benefit under reference to an EFSA guidance document published in 2016.
Call for harmonized use of the term ‘probiotics’
According to IPA Europe, ‘probiotics’ is therefore not a health claim and does not require authorization under the EU Claims Regulation. At the same time, it emphasizes the need for clear rules on the use of the claim as this contributes to a fair competitive environment for food businesses and helps consumers to make informed choices. In December 2023, IPA Europe therefore called for a clearly defined framework for the use of the term ‘probiotics’ in the EU. IPA Europe recommends the following four criteria for consistent use thereof:
- characterization of the species level and identification of at strain level;
- the probiotic strain must be safe for the intended use, e.g. based on the QPS list;
- the probiotic status should be scientifically documented; and
- the probiotic strains must be alive in the product and in a sufficient amount up to the end of shelf-life.
Final comment
The EU knows a fragmented regulatory landscape when it comes to the use of the term ‘probiotics’. Despite IPA Europe’s efforts for a harmonized approach throughout the EU, we will need European legislation or a ruling from the European Court of Justice to have the same rules in all EU member states. For now, the term ‘probiotics’ is (fortunately) not completely banned in the Netherlands and neither in quite a few other EU Member States.
This blogpost has also been published in Dutch at VMT.nl.
Posted: December 14, 2023 | Author: Karin Verzijden | Filed under: Food |
 Last month, the conference Regulating the Future of Foods took place in Barcelona, gathered more than hundred professionals active in the fields of precision fermentation and cellular agriculture. The purpose of the conference was to define hurdles and investigate opportunities in the current regulatory framework applicable to this sector. Many interesting presentations took place discussing the global perspective of our future food system and AXON moderated a workshop targeting tastings of cultivated foods, formulating a number of conversation topics. In this blogpost, we share the outcome of the discussions that took place during this workshop.
Last month, the conference Regulating the Future of Foods took place in Barcelona, gathered more than hundred professionals active in the fields of precision fermentation and cellular agriculture. The purpose of the conference was to define hurdles and investigate opportunities in the current regulatory framework applicable to this sector. Many interesting presentations took place discussing the global perspective of our future food system and AXON moderated a workshop targeting tastings of cultivated foods, formulating a number of conversation topics. In this blogpost, we share the outcome of the discussions that took place during this workshop.
Regulatory frameworks for tastings
Tastings of these products already took place, for instance in Israel. So far however only two countries have developed a legal framework for this purpose, notably Singapore and the Netherlands. In the tastings workshop, the procedure and the data requirements for setting up tastings in these countries have been explained, as you can see in the powerpoint inserted below. Furthermore, the following items were discussed.
1. Should the SGP / NL template become the blueprint for tastings or rather do this under the radar?
As follows from the comparison of the regulatory frameworks in Singapore and the Netherlands, these hugely overlap. Therefore, the question arose if these frameworks should become the blueprint for tastings in other countries. Kind Earth.Tech rightfully pointed out that at the beginning of the cultivated meat industry 10 years ago, all tastings held were illegal. They were however important to demonstrate proof of principle and to create appetite for further research. At the current state of the industry, all participants in the workshop however favoured a framework for tastings. Especially for start-ups, a tastings framework is valuable for showing both press and investors what they are up to. Furthermore, the Dutch initiative is useful to convince other EU Member States to develop similar initiatives. Bluu Seafood pointed out that Germany in particular was pretty shy to do so, as it considered tastings not to be in compliance with the EU Novel Food Regulation. Now German start-ups can point to the Dutch framework and request their authorities to take such initiative.
Another reason why tastings might be useful, is that the regulatory process takes a substantial amount of time. In Singapore, predicted timelines for Novel Food approval are between 9 – 12 months, but in reality, these go up. In fact, we have not seen any approvals for cell-based meat products since those for Good Meat and Upside Foods in Q4 2020. In the EU, the theoretic term for Novel Food approvals is about 18 months, but we know from practice it is more realistic to count 2 – 3 years. For the new industries of cellular agriculture, it remains to be seen if this term will apply as well. Demonstrating proof of principle during tastings can be a welcome deliverable before the final go.
Both in Singapore and the Netherlands, tastings should be done in a confined area, not open to the general public. This was understood by all participants in the workshop. At the same time, the concern was expressed that tastings should not become too clinical. They are meant to enjoy food products after all, not to evaluate medicinal products. Provided that a confined area and a selected audience can guaranteed, they can also be set up in a restaurant. Organising tastings during regular opening hours of restaurants will not yet be feasible, as such would fall within the scope of “placing on the market” under article 3.8 of the EU General Food Law Regulation. And placing on the market of Novel Foods requires pre-market approval.
2. For those previously involved in tastings: are they worth the efforts and did they bring you the data you were looking for?
The parameters set in the regulatory tasting frameworks are meant to be high enough to ensure food security but not dissuasive for companies to demonstrate safety prior to obtaining market authorization. Nevertheless, it takes considerable efforts to accommodate these parameters. For instance, producing useful microbiology data requires a minimum quantity of the product to be produced, which at this stage is still very expensive.
Obviously, the tastings themselves require substantial product input, often in combination with various non-cultivated carriers creating a hybrid product. This requires investments which for start-up companies can be challenging.
However, tastings have proved most valuable to collect input from chefs who will be working with cultivated products and to provide proof of principle to investors. In particular, repeating tastings with the same chefs offers the advantage that they can monitor and comment progress made and provide suggestions for further improvement, especially to accommodate the local palette.
2. Is the available guidance from the regulators sufficiently clear to decide how to set up the tasting and establish the safety of your product? Has the regulator been helpful in clarifying any company queries?
Companies that already conducted tastings in Singapore considered the Singapore Food Authority (SFA) to be helpful in not only overseeing this process, but also in facilitating it. Where a first tasting takes quite some work in setting up safety documentation and addressing any potential concerns of the regulator, repeated tastings proved to require much less preparations. This is particularly true if all tastings take place at the same location, as it facilitates the medical contingency plan. This requires the indication how much time it takes to reach a medical centre if any problems occur.
Most importantly, permission for tastings being granted is perceived as a quiet vote of confidence by the regulator, as it seems to have a minimum level of comfort with the safety of the product to be tasted.
3. Do you see any value in tastings to be set up at an EU level as opposed to national level only?
No, not really. It is important to create a product that appeals locally, and this may differ from community to community. Also, it was discussed that tastings organized at a central level could induce companies to rely on the greatest common denominator, i.e. a burger. This would in fact be a pity, as cultivated foods offer so much more opportunities than that. This was for instance demonstrated by Vow Foods who made a crème brûlée with its cultivated quail.
4. Any ideas what could be done to prevent that tastings slow down regulatory approvals?
The representative from SFA explained that the total staff available for the safety assessment of Novel Food AND the evaluation of an exemption for the tasting and sensory evaluation of Novel Foods is four to five persons. It is easy to understand that the more time is spent on the evaluation of tastings, the less time can be spent on the safety assessment of Novel Foods as such.
It was discussed that if tastings go wrong, for instance by creating a health problem, they imply a risk, not only for the product at stake, but also for the sector. On the other hand, tastings also spark enthusiasm, as was demonstrated by Solar Foods who even held tastings after obtaining market approval in Singapore.
So the communis opinion was that tastings should be dosed. Do not overdo it, but carefully weigh the advantage it can bring your company at a particular moment of its lifetime, for example during a funding round.
5. Do tastings qualify as studies under the EU Transparency Regulation, that should be notified to EFSA during an application for authorization of a Novel Food?
The EFSA representative present in Barcelona was quite outspoken regarding this question. If you have any concerns about toxicity or any other possible hazard, you do not want to expose your intended audience for tastings to such hazard. A tasting session is not a study. So no, tastings as such do not have to be notified to EFSA under the Transparency Regulation.
What’s next?
In the Netherlands, the CAN Expert Committee is expected to be nominated early 2024 and local companies are already gearing up for organizing tastings in situ. So soon the Dutch will continue to work on their Dutch dream. Watch that space!
Posted: December 8, 2023 | Author: Jasmin Buijs | Filed under: Food |
 Germany’s highest court, the Bundesgerichtshof, asked the European Court of Justice (ECJ) this summer to explain the use of ‘on hold’ claims for so-called ‘botanicals’. The question is whether these substances may be advertised with health claims or general, non-specific health benefits as long as the assessment by EFSA has not been completed and the European Commission has not yet taken a final decision on the authorization of these claims.
Germany’s highest court, the Bundesgerichtshof, asked the European Court of Justice (ECJ) this summer to explain the use of ‘on hold’ claims for so-called ‘botanicals’. The question is whether these substances may be advertised with health claims or general, non-specific health benefits as long as the assessment by EFSA has not been completed and the European Commission has not yet taken a final decision on the authorization of these claims.
This question stems from proceedings initiated by the German unfair competition association Verband Sozialer Wettbewerb e.V.. We owe many ECJ rulings to this German association, including the famous TofuTown ruling from which it follows that milk designations are reserved for animal dairy products only. We covered the TofuTown case in an earlier blogpost. The current case is against the food supplement company Novel Nutriology, which sells, among other things, an ‘anti-stress’ supplement containing saffron and melon juice extract.
The saffron extract is said to create a more positive mood, according to the seller’s website. This expression is backed by the results of an open study on 50 participants over a 30-day period. Research has shown that the melon juice extract reduces feelings of stress and fatigue, according to the website. Verband Sozialer Wettbewerb e. V. considers these to be unpermitted health claims. It therefore requested Novel Nutriology to stop making these claims, but the supplement company did not listen.
Approved health claims
Health claims are in principle only allowed if they are included in the lists of approved claims under the EU Claims Regulation. This follows from article 10(1) Claims Regulation. References to general, non-specific health benefits such as ‘heart health’ and ‘mental energy’ are allowed if accompanied by a specific, approved claim (article 10(3) Claims Regulation). However, health claims for botanicals are ‘on hold’ and are therefore not included in the lists of approved claims. Therefore, the literal text of Article 10(1) and (3) Claims Regulation cannot be met when making health claims for botanicals.
Claims Regulation nevertheless applicable to botanicals?
To determine whether Novel Nutriology violates the prohibition on making unauthorized health claims, it is essential to know whether Article 10(1) and (3) of the Claims Regulation apply to botanicals. If not, violation of these provisions is out of the question.
As can be seen from the summary of the request for the preliminary ruling, the view is generally taken that references to general health benefits of botanicals must comply with Article 10(3) Claims Regulation. The requirements of this provision are met if the generic health benefit is accompanied by the full, specific health claim that is ‘on hold’. That full ‘on hold’ claim, although not officially authorized, may be used in accordance with the transitional measures in Article 28(5) and (6) Claims Regulation. It would not be compatible with the purpose of Article 10(1) and (3) Claims Regulation to exclude these provisions entirely for botanicals. Such would namely mean that botanical substances may be advertised with non-specific health claims without a scientific assessment of the specific claim supporting them.
Alternative views
Nevertheless, the Bundesgerichtshof finds it unclear whether Article 10(1) and (3) Claims Regulation apply to botanicals. As an argument against applicability, the German court states that the Union legislator would have considered an absolute ban on general health benefits as too broad. Therefore, the Union legislator intended to ban such only if the general claim is not accompanied by an approved, specific health claim.
It is currently however impossible to obtain approval for botanical claims that are ‘on hold’ due to the European Commission’s inaction. By making Article 10(3) Claims Regulation applicable to these substances, the prohibition becomes broader than the Union legislator would have intended. It should therefore be assumed that general health benefits for botanicals are not regulated until the European Commission continues the authorization procedure for ‘on hold’ claims. Based on this alternative view, Article 10(3) of the Claims Regulation should not apply until then.
A second argument raised against the applicability of Article 10(1) and (3) Claims Regulation to botanicals is that the European Commission has not taken action on ‘on hold’ claims for many years. Upholding the applicability of Article 10(1) and (3) Claims Regulation, which cannot be met for ‘on hold’ claims, would lead to a disproportionate restriction of the interests of companies making such claims.
Previous ECJ rulings
 Whether the aforementioned alternative views are sufficient to exclude botanicals from the scope of Article 10(1) and (3) Claims Regulation remains to be seen. As highlighted in a report published last September, companies are able to make health claims for botanicals that are included in the ‘on hold’ list under the transitional regime of Article 28(5) and (6) of the Claims Regulation. Moreover, the ECJ ruled in previous cases that companies making ‘on hold’ claims are not disproportionately disadvantaged. Since they can make claims without EFSA having assessed these and/or without a final decision from the European Commission, they are in fact favored.
Whether the aforementioned alternative views are sufficient to exclude botanicals from the scope of Article 10(1) and (3) Claims Regulation remains to be seen. As highlighted in a report published last September, companies are able to make health claims for botanicals that are included in the ‘on hold’ list under the transitional regime of Article 28(5) and (6) of the Claims Regulation. Moreover, the ECJ ruled in previous cases that companies making ‘on hold’ claims are not disproportionately disadvantaged. Since they can make claims without EFSA having assessed these and/or without a final decision from the European Commission, they are in fact favored.
Aforementioned report also highlights that the ECJ previously made explicit that Article 28(5) Claims Regulation makes an exception for the use of specific health claims as referred to in Article 13(1)(a) Claims Regulation that have not yet been officially approved. The report therefore concludes that the mentioned transitional regime only applies to the full specific claim and not to general health benefits. General health benefits would therefore not be allowed, although it is recognized that some EU member states do accept general health benefits for botanicals when accompanied by the full ‘on hold’ claim.
Relevance for practice
Health claims for botanicals are currently accepted in the Netherlands if the roadmap of the Dutch agency regulating health products (the Keuringsraad) in cooperation with the NVWA, is met (see here, in Dutch). In brief, this means that (i) the substance and claim are present on the ‘on hold’ list, (ii) potential conditions of use such as a daily dosage are included on the label, (iii) the claim is in line with the wording accepted on KOAG KAG’s ‘indicative list’, and (iv) a disclaimer regarding the ongoing approval procedure is made.
If the ECJ rules that Article 10(1) and (3) Claims Regulation does not apply to botanicals, it is uncertain whether the Keuringsraad roadmap can be upheld. It is however more likely that the ECJ will clarify that botanicals do fall within the scope of the aforementioned provisions. In that case, various outcomes are still possible. In connection with the transitional provisions of Article 28 Claims Regulation, a difference may arise for full ‘on hold’ claims and general health benefits. Such will partly depend on whether the ECJ sticks to the literal text of the transitional provisions (which would prohibit the use of general health benefits), or takes into account current practices (in which case advertising with general health benefits remains allowed under certain conditions). To be continued!
Posted: October 23, 2023 | Author: Karin Verzijden | Filed under: Authors, Food, New Genomic Techniques, organic |
 Genetically modified organisms (GMOs) do not generally receive a warm welcome from the average EU citizen. Possibly this is a case of ‘unknown makes unloved’. But GMOs can also bring positive things, for example, in the field of plant breeding. This summer, the European Commission published a proposal for an NGT Regulation. What exactly does this proposal entail and what are the expected consequences in practice? And how was this proposal received by the European Parliament in its draft report of 16 October last in the first reading of the legislative procedure?
Genetically modified organisms (GMOs) do not generally receive a warm welcome from the average EU citizen. Possibly this is a case of ‘unknown makes unloved’. But GMOs can also bring positive things, for example, in the field of plant breeding. This summer, the European Commission published a proposal for an NGT Regulation. What exactly does this proposal entail and what are the expected consequences in practice? And how was this proposal received by the European Parliament in its draft report of 16 October last in the first reading of the legislative procedure?
Potential benefits GMOs and reluctance
GMOs can help develop plants that are more drought tolerant or less susceptible to certain fungi. This reduces the need for pesticides in cultivation, precisely one of the objectives of the EU Farm-to-Fork strategy that the Commission published at the beginning of its mandate in May 2020. At the same time, in the Netherlands the proposal for the NGT Regulation prompted Odin, Demeter, Ekoplaza and Greenpeace, among others, to start a petition calling for “Keep our food genetically modified free.” (in Dutch: Houd ons voedsel gentech vrij). At the time of writing this blogpost, the petition counts 47.709 signatories.
Scope of application NGT Regulation
The intended Regulation applies to NGT plants and to NGT products, meaning food and feed containing or consisting of or produced from NGT plants and other products containing or consisting of such plants. NGT plants are obtained from the following two genomic techniques or a combination thereof:
- Targeted mutagenesis: this is a technique that results in changes to the DNA sequence at precise locations in an organism’s genome;
- Cisgenesis: this is a technique that results in the insertion, into the genome of an organism, of genetic material already present in the total genetic information of that organism or another taxonomic species with which it can be crossed.
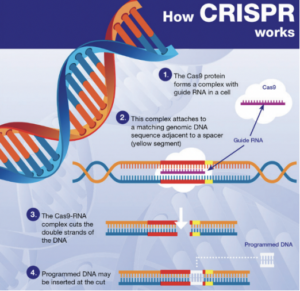
In essence, NGTs are genomic techniques such as CRISPR-Cas9. These techniques result in more targeted genomic modifications than older genomic techniques, which often involve the introduction of heterologous genetic material. The European Commission therefore recognizes that any risks associated with the use of NGTs are lower than those associated with older genetic techniques. This is what the risk assessment in the draft Regulation specifically addresses.
Two specific regulatory regimes for category 1 and category 2 NGT plants
- Category 1 NGT plants: these plants are considered equivalent to conventionally bred plants based on the criteria in Annex I
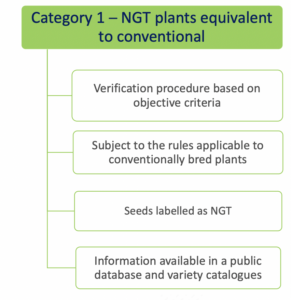 of the NGT Regulation and as such do not need to undergo complete GMO risk assessment. Instead, a notification to a national GMO authority or to EFSA is sufficient;
of the NGT Regulation and as such do not need to undergo complete GMO risk assessment. Instead, a notification to a national GMO authority or to EFSA is sufficient;
- Category 2 NGT plants: these plants are considered as GMOs and as such are subject to the GMO rules for authorization, traceability and labeling, however according to a modified system of more targeted risk analysis.
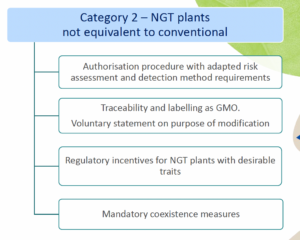 In addition to NGT plants, these regulations also apply to NGT products: that includes foods with ingredients made from such plants. This is why this proposed Regulation is so relevant for innovative food business operators.
In addition to NGT plants, these regulations also apply to NGT products: that includes foods with ingredients made from such plants. This is why this proposed Regulation is so relevant for innovative food business operators.
As mentioned above, for category 1 NGT plants, a notification procedure takes place and for category 2 NGT plants a full swing risk assessment. Both procedures involve a substantive assessment of whether plant material produced using targeted genetic techniques complies with the specifically developed rules for risk assessment. It is expected that the procedure for category 1 NGT products can be completed within a year, while the permit process for category 2 NGT foods is actually identical to that for regular GMOs. An innovation is that the NGT Regulation provides certain incentives for specific category 2 NGT foods that should speed up or simplify the assessment process.
Transparency regarding genetic engineering
Producers of organic products and advocacy organizations are concerned that, based on the NGT Regulation, genetically engineered food is walking into stores undetected. But is this concern justified? Based on the current text of this Regulation, both category 2 and even category 1 NGT products are excluded from organic production. Indeed, in the consultation process leading up to the NGT Regulation, the freedom of choice of consumers to buy food containing or not containing GMOs appeared to be an important issue. At the same time, the European Parliament considers the prohibition for organic farmers to use conventional-like category 1 NGT’s neither science-based, nor politically justifiable. It therefore calls in its draft report to create a fair level playing field and to only ban category 2 NGT plant material from organic production.
Category 1 NGT products listed in public database?
Whether or not allowed in organic production, there is no specific labeling requirement for Category 1 NGT products (Q 11 from EC Q&A). However, plant material with Category 1 NGT status must be included in a public database, expected to be similar to the Novel Food consultation database. In the Commisson’s proposal, plant reproduction material with Category 1 NGT status must additionally be labeled as such, with the identification number of the plant from which it is derived. At the level of production, this would provide an extra safeguard for distinguishing between conventionally grown crops and crops in which genetic engineering has been used. The European Parliament is however critical about this additional requirement in its draft report. It considers such discriminatory since category 1 NGT plants are conventional-like. According to the European Parliament, transparency and consumer choice are sufficiently ensured by disclosure in a public database. Therefore, even if the modifications proposed by the European Parliament will stand, there is no reason to believe that foods produced using CRISPR-Cas9 could go completely unnoticed.
Conclusion
With current changes in climate and constraints on available agricultural land with a growing world population, plant harvests will come under increasing pressure. NGTs are expected to meet the need to better equip plants for such challenges. The proposed NGT Regulation aims to simplify and thereby accelerate market access for such technology. For category 1 NGT products, except for a few open ends, this premise seems feasible in practice. A huge improvement with respect to current legislation is that the nature of the genetic change is decisive for risk evaluation, not the technique by which it is produced (product-based vs. process-based approach). For category 2 NGT products, market access is not expected to become much simpler based on the current proposal. Nevertheless, this proposal looks hopeful for plant innovations and thus for our food products of tomorrow. Also, there seems to be sufficient transparency to distinguish between crops produced with and without genetic engineering. My very bald hope would be that based on positive experiences with this regulation, the scope of this regulation will be extended to other fields, such as cellular agriculture and/or fermentation-based products. We will continue to follow closely how this legislation will develop.
Source images
- How CRISPR works: EU Parliament briefing on Plants produced by new genomic techniques
- Category 1 NGT plants: PPT DG Santé on Farm to Fork Strategy presented during EFFL Conference on 19 October 2023.
- Category 2 NGT plants: idem (2)
Thanks to my colleagues Jasmin Buijs and Max Baltussen for their valuable feed-back.
Posted: June 21, 2023 | Author: Karin Verzijden | Filed under: Authors, Food, novel food |
 On May 25, the European Court of Justice (ECJ) ruled in a dispute arising between two manufacturers of food supplements. This is the first decision on the interpretation of the current Novel Food Regulation (applicable since Jan. 1, 2018). The dispute concerned the method of production of the functional ingredient spermidine. The dispute was hoped to clarify the question of what exactly a “new production process” under the Novel Food Regulation entails. The decision follows a request from a court in Austria, where this answer was needed to resolve a national dispute. This answer is also relevant to other countries, as the Novel Food Regulation applies EU-wide.
On May 25, the European Court of Justice (ECJ) ruled in a dispute arising between two manufacturers of food supplements. This is the first decision on the interpretation of the current Novel Food Regulation (applicable since Jan. 1, 2018). The dispute concerned the method of production of the functional ingredient spermidine. The dispute was hoped to clarify the question of what exactly a “new production process” under the Novel Food Regulation entails. The decision follows a request from a court in Austria, where this answer was needed to resolve a national dispute. This answer is also relevant to other countries, as the Novel Food Regulation applies EU-wide.
Why do people consume spermidine?
Spermidine supplementation takes place with a view to supporting cellular autophagy, or cell renewal. This could promote the prevention of cardiovascular disease, prevent food allergies, and control the symptoms of diabetes. It has even been suggested that spermidine could extend human lifespan by 5 to 7 years. So says Advocate General (A-G) Campos Sánchez-Bordona in his opinion on the case dated Jan. 19 this year, citing various scientific sources (see footnotes 12 and 13 of this opinion). The A-G is an important advisor to the ECJ, which generally follows his or her opinions.
Background to this dispute.
The dispute in Austria was brought by The Longevitiy Labs (TLL), which markets the spermidine supplement spermidineLIFE. TLL extracts spermidine from ungerminated wheat germs through a complex and expensive chemical process. TTL applied for and obtained an EU Novel Food authorization for this food product (see Union List, entry “spermidine-rich wheat germ extract“). Then competitor Optimize Health Solutions enters the market with its own spermidine supplement. This is produced using a much simpler and therefore cheaper production process based on hydroponic cultivation of buckwheat seeds in an aqueous solution with synthetic spermidine. After harvesting the seedlings are washed in water, dried and milled to obtain flour. TLL believes that Optimize Health also needs a Novel Food authorization to market its product. It instituted proceedings seeking an injunction against further marketing of Optimize Health’s product without such an authorization.
Optimize Health argues it does not need a Novel Food authorization, because its product is not covered by the Novel Food Regulation. It is a fully dried traditional food obtained without any selective novel extraction method. Furthermore, it states spermidine has been available in food supplements in the EU market for more than 25 years. The germination of the buckwheat seeds of which its product is made would be a primary production process, to which the EU Hygiene Regulation applies, not the Novel Food Regulation. Furthermore, the Novel Food Regulation does not apply because its product involves “plants prior to harvesting” and these do not count as food under the EU General Food Law Regulation. The Austrian court decided that clarification of European law was needed to resolve this dispute and referred five questions to the ECJ.
Questions from the Austrian referring court
National judges who refer questions to the ECJ usually go for as many anchors as possible and thinking three steps ahead. If a possible answer by the ECJ to one question leads to a follow-up question by the inquiring national court, that follow-up question will be submitted upfront as well. The downside of this system is that if answering the first question is the end of the matter, answering the remaining questions is no longer needed. That is what we call procedural economy. In summary, the referring Austrian court asks the following questions:
- Should a food consisting of flour from buckwheat seeds with a high spermidine content be qualified as Novel Food in the category “foods isolated from (parts of) plants?”
- If not, might it be a Novel Food because a new production process has been used and does that term include primary production processes?
- If it is a novel production process, does it matter whether that process was not applied at all or only not applied to spermidine?
- If primary production processes are not covered by the term “new production process”, is it correct that the process of germinating buckwheat seeds in a nutrient solution containing spermidine is not covered by the Novel Food Regulation because it does not apply to plants prior to harvesting?
- Does it make a difference whether the nutrient solution contains natural or synthetic spermidine?
Decision of the ECJ
The ECJ answers question 1 – be it with some reservation – in the affirmative. Optimize Health’s product is a Novel Food because there is no evidence that this product was used for human consumption to any significant degree within the EU before 1997. This short answer is somewhat disappointing, as it makes answering the remaining questions irrelevant. Still, the ECJ does share two interesting considerations regarding the current Novel Food Regulation. It points out that the concept of “history of safe use within the Union” is not defined with respect to Novel Foods that must undergo the full authorization process. However, it is defined with respect to traditional foods from third countries. These are products that have been used as food outside the EU for considerable time, such as chia seeds. These products are subject to the requirement that the safety of the food has been confirmed by compositional data and experience of continued use for at least 25 years in the usual diet of a significant number of people in at least one third country. The ECJ finds that this requirement must be applied by analogy to the spermidine in question and concludes that said data have not been provided.
Propagation methods vs. complete production process
Another consideration of the ECJ concerns the Novel Food category of “food isolated from (parts of) plants”. An exception applies to products made by non-traditional propagation methods, which do not result in significant changes in the composition or structure of the food in question. The ECJ ruled that a distinction must be made between:
(1) propagation processes of which the purpose is to produce new plants; and
(2) processes involving the entire production process of a food product.
The process applied to Optimize Health’s product to achieve a high spermidine content falls into the second category. In other words, a manufacturing technique to enrich a product is not the same as a propagation technique. The ECJ instructs the Austrian court to take this into account when deciding the case at the national level. This reduces the likelihood that the exception to the Novel Food category above will apply and puts the ECJ’s reservation into perspective. Good chance, therefore, that the Austrian national court will indeed determine that Optimize Health’s product is a Novel Food.
New production procedure according to the A-G
With the above answer, the question of the Austrian court has been answered and the ECJ does not get to the remaining four questions. It is of course unfortunate that we will not know the ECJ’s decision on this. Therefore, it is interesting to see how the A-G ruled on this. Well: according to the A-G, enriching buckwheat seeds with spermidine should be considered a new production process. He argues that bio-enrichment with spermidine changes the composition and nutritional value of the buckwheat seeds flour. Indeed, its spermidine content becomes 106 times higher than that of un-enriched buckwheat seeds. The A-G cites studies according to which a higher spermidine content may be beneficial to health, but which also indicate that too high an amount of spermidine could be harmful to cells.
The A-G therefore concludes that prior authorization of Optimize Health’s product is indispensable to ensure food safety and avoid risks to consumers. He also refers to products such as selenium-enriched mushrooms and mushrooms treated with ultraviolet light after harvest to increase their vitamin D2 content, where such authorization has also taken place. Furthermore, the A-G argues that the effects and thus the safety of a new production process should be assessed in each individual case and thus not in general. The same production process may affect one foodstuff differently from another.
Conclusion
This spermidine case clarifies the criterion that should be applied to determine whether a food has a history of safe use in the EU and thus qualifies or not as Novel Food. By the way, this is not entirely new – there has already been a guidance document from the European Commission “Human Consumption to a Significant Degree” since 1997 that argues essentially the same thing. However, when this judgment is considered in conjunction with the A-G’s opinion, it does provide relevant new information for determining how to establish whether there is a new production process.
This is the case if it is established that an applied process significantly alters the composition and nutritional value of a foodstuff compared to a foodstuff to which such a process has not been applied. Thus, in such a case, a food must obtain authorization under the Novel Food Regulation. The A-G recognizes that the question whether it is relevant if the production process has been previously applied to any foodstuff (rather than to the foodstuff specifically) cannot simply be answered based on the legal text. So that requires interpretation of the specific article of the Novel Food Regulation on new production processes in the light of its context and purpose. For now, we do not yet know whether the ECJ supports his interpretation. Hopefully we will find out in another case. It does sound plausible to me.
Posted: April 21, 2023 | Author: Jasmin Buijs | Filed under: Authors, Food, Information |
 The ‘farm to fork’ strategy calls for better communication to consumers about healthy and sustainable foods. To make this happen, EU legislation on food information to consumers is currently being revised.
The ‘farm to fork’ strategy calls for better communication to consumers about healthy and sustainable foods. To make this happen, EU legislation on food information to consumers is currently being revised.
The revision of the FIC Regulation covers front-of-pack nutrition labeling, establishment of nutrient profiles, origin labeling and date marking (‘best before’ / ‘use by’). The revision as prompted by the ‘farm-to-table’ strategy further relates to the labeling of alcoholic beverages, as announced in the European Cancer Control Plan.
Need for change
Our daily nutritional intake in Europe is not in line with national and international dietary recommendations. This leads to diet-related chronic diseases, such as diabetes and cardiovascular disease, with all kinds of consequences. According to the European Commission, this is partly because current labeling rules do not provide sufficient guidance for consumers to choose healthy foods.
Various proposals
As part of the legislative process, the European Commission published a so-called ‘Impact Assessment‘ at the end of 2020. The Impact Assessment sets out the labeling rules that need adjustment, and the different options for amendment as proposed by the European Commission. The Impact Assessment for tightened regulations on alcoholic beverages labeling was published later, in the summer of 2021. Below we discuss which changes to the FIC Regulation are proposed in the Impact Assessments and why, and touch upon the options presented for each topic. Later this year, the European Commission will follow up with a proposal for an amended regulation.
FOP labeling
Consumers do not always understand the nutritional information on packaging, which makes it difficult to choose healthy foods. To help consumers making better decisions, there are all kinds of voluntary initiatives for clear front-of-pack (FOP) nutrition labeling. Examples include the much discussed Nutri-Score, but also other initiatives such as the traffic light system (UK), the keyhole (Scandinavia) and the battery (Italy). However, these diverse FOP logos do not necessarily help European consumers as they do not provide equal access to information. The European Commission is concerned that this could lead to fragmentation of the single market, costs for food companies operating in several member states, and confusion among consumers. Harmonizing FOP logos and making them mandatory or not, are options on the table.
 Nutrient profiles
Nutrient profiles
Another proposed change concerns nutrition and health claims on unhealthy products. For example, consider the claim “source of fiber” for biscuits and “rich in vitamin C” for soft drinks. Such claims obscure the unhealthy profiles of these products, which leads to ‘health washing’ (term as introduced by the Dutch consumer association, see here). Nutrient profiles, i.e. thresholds for fats, sugars and/or salt above which the use of nutrition or health claims is restricted, could provide a solution to this. Such thresholds could help preventing consumer deception and create a level playing field for food companies. According to the Claims Regulation, nutrient profiles should already have been established as early as 2009, but this never materialized due to intense debate on the matter. The revision of the FIC Regulation revitalized this discussion. Based on stakeholder feedback on the Impact Assessment, the time seems ripe for change now.
Origin labeling
There is a growing demand from consumers to know the origin of food products. This allows consumers to make more sustainable choices, such as by choosing local products. Origin labeling is already mandatory for certain types of meat, among others. In the absence of harmonized rules regarding other food categories, a number of member states developed national rules for this purpose. Rules differentiating from member state to member state however lead to unequal access to information within the EU and fragments the internal market. The European Commission is therefore investigating whether and, if so, how, European rules on origin labeling could be further extended. This could mean an expansion of the product groups for which origin indication is required. Current discussions also include the production phase to which the indication should refer and the area size referred to (EU / non-EU, or e.g. country or regional level).
Date marking
We currently have two types of date marking. Whereas ‘use by’ refers to the expiry date for food safety reasons, ‘best before’ refers to the date by which the food retains its optimum quality. After this date, color variations may occur, for example, but the product is still safe to eat. The Impact Assessment states that less than one in two consumers understand the meaning of the two date markings. As a result, a lot of food ends up in the waste bin unnecessarily. By estimate 10% of the 88 million tons of food that is wasted annually is linked to date marking. In the context of improving sustainability by reducing food waste, the Impact Assessment presents options to educate consumers about date marking. It also suggests the possibility of removing the ‘best before’ quality date where this has little or no added value.
 Labeling of alcoholic beverages
Labeling of alcoholic beverages
Health damage from alcohol is a serious public health issue in the EU. To reduce alcohol consumption, it is important to inform consumers about what is actually in alcoholic beverages, both in terms of ingredients and nutrients. Currently, this is not mandatory for alcoholic beverages with an alcohol volume above 1.2%. Having said that, there are various initiatives for better information provision on alcohol, such as self-regulation within the beer and spirits sectors. To create a level playing field for operators, the European Commission communicated among others a proposal to remove the aforementioned labeling exception for all alcoholic beverages. Some of the then required information could be communicated off-label via a QR code.
Follow-up steps
Interested parties were able to share feedback with the European Commission after the publication of the two Impact Assessments. To further prepare the legislative proposal, a public consultation took place from 13 December 2021 to 7 March 2022. This process was designed to gather further views, experiences and suggestions on food labeling from stakeholders. As many as 3224 citizens, companies, interest groups, public authorities and other parties filled out the questionnaire made available for this purpose. A large proportion of respondents expressed their support for a harmonized FOP logo, as well as for improved terminology or a visual presentation of date marking. The European Commission’s legislative proposal is expected later this year.
This blogpost has also been published in Dutch at VMT.nl. The author thanks Marie-Claire Evers for her translation of this blogpost into English.
Posted: January 3, 2023 | Author: Jasmin Buijs | Filed under: Authors, Food |
 Last October, AXON contributed to the 16th European Food and Feed Law Conference by a session on circular economy, waste, packaging law, alternative materials, and the Single-Use Plastics (SUP) Directive. While we were back then still waiting for proposals by the European Commission on packaging (waste) and bioplastics, these long-awaited proposals have now been published. This blogpost discusses the main take-aways from these recent European proposals and provides deeplinks to the texts involved.
Last October, AXON contributed to the 16th European Food and Feed Law Conference by a session on circular economy, waste, packaging law, alternative materials, and the Single-Use Plastics (SUP) Directive. While we were back then still waiting for proposals by the European Commission on packaging (waste) and bioplastics, these long-awaited proposals have now been published. This blogpost discusses the main take-aways from these recent European proposals and provides deeplinks to the texts involved.
Packaging problem
Packaging plays without a doubt a very important role in the placing on the market of food. It protects and preserves food, and therefore contributes to increased shelf life and reduced amounts of food waste. It also offers a way to communicate food information to the consumer. At the same time, packaging, just like anything else we create, leaves an impact on the environment. As communicated in the Green Deal, it is the EU’s ambition to lower our amount of packaging waste as part of the green transition.
Bio-plastics
In a search to meet the EU’s goals for a circular economy and climate-neutrality by 2050, bio-plastics are emerging on the market as alternatives for conventional plastics. Bio-plastics can bring several advantages such as making packaging production less dependent on fossil fuels and reducing litter as they may dissolve over time. Their use is however not without challenges as discussed in an earlier blogpost. To improve the understanding around these materials and to clarify where bio-plastics can bring genuine environmental benefits, the European Commission recently published a Communication towards other EU bodies on a policy framework for bio-plastics. As is already clear from existing rules on environmental claims, the Commission stresses that generic claims on packaging such as ‘bio-plastic’ should be avoided to stay away from greenwashing and misleading consumers. It furthermore proposes to only label bio-plastic packaging and other products as ‘biobased’, ‘biodegradable’ or ‘compostable’ when it meets certain conditions. The main take-aways for businesses are as follows:
dissolve over time. Their use is however not without challenges as discussed in an earlier blogpost. To improve the understanding around these materials and to clarify where bio-plastics can bring genuine environmental benefits, the European Commission recently published a Communication towards other EU bodies on a policy framework for bio-plastics. As is already clear from existing rules on environmental claims, the Commission stresses that generic claims on packaging such as ‘bio-plastic’ should be avoided to stay away from greenwashing and misleading consumers. It furthermore proposes to only label bio-plastic packaging and other products as ‘biobased’, ‘biodegradable’ or ‘compostable’ when it meets certain conditions. The main take-aways for businesses are as follows:
Biobased:
- specify the exact and measurable share of biobased plastic content in the packaging; and
- ensure that the biomass is sustainably sourced – priority should be given to the use of organic waste or by-products rather than to primary biomass.
Biodegradable:
- specify that biodegradable packaging should not be littered;
- do not label products covered under the SUP Directive (the scope of which we discussed in more detail here and here) and other short-lived applications and/or litter-prone packaging as biodegradable; and
- specify how long the product needs to biodegrade, under which circumstances and in what environment.
Compostable:
- label only industrially compostable plastics that comply with relevant standards – the Commission will request the revision of the European Standard EN 13432:2000 for this purpose;
- use industrially compostable plastics only if the environmental benefits are higher than their alternatives and if they do not have a negative impact on the quality of the compost, taking into account consumer behavior; and
- specify the way in which the packaging should be disposed of using pictograms.
The Communication on bio-plastics refers, where relevant, to the Commission proposal for a Packaging and Packaging Waste Regulation (PPWR). The aforementioned proposal was also published at the end of last year and is discussed next.
Packaging and Packaging Waste Regulation
Before diving into the Commission’s proposal for a PPWR, it is useful to provide some background on the legal framework for packaging waste as guided by the Waste Framework Directive (WFD).
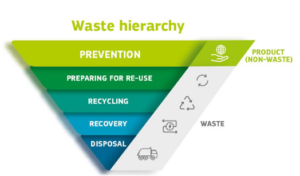 The WFD (currently under revision) introduces the waste hierarchy for waste management, which establishes an order of the preferred disposing route. Waste should in the first place be prevented/reduced. If this is not possible, re-use and thereafter recycling options should be looked into. Only in case this is (or is no longer) an option, energy recovery through incineration or ultimately landfill disposal should be considered. To reduce waste, the WFD also introduces the polluter pays principle and the extended producer responsibility, based on which the waste producer bears financial and/or organizational responsibility for the management of the waste stage at the end of a product’s life cycle.
The WFD (currently under revision) introduces the waste hierarchy for waste management, which establishes an order of the preferred disposing route. Waste should in the first place be prevented/reduced. If this is not possible, re-use and thereafter recycling options should be looked into. Only in case this is (or is no longer) an option, energy recovery through incineration or ultimately landfill disposal should be considered. To reduce waste, the WFD also introduces the polluter pays principle and the extended producer responsibility, based on which the waste producer bears financial and/or organizational responsibility for the management of the waste stage at the end of a product’s life cycle.
The proposal for the PPWR is the Commission’s answer to the revision of the current Packaging and Packaging Waste Directive (PPWD), which focuses on reducing, re-using and recycling packaging. The choice of legal instrument (a regulation rather than a directive) should facilitate a harmonized approach across the various EU Member States. The Regulation is however not an easy read. Although it contains only 65 legal articles, it includes many exceptions to the measures it proposes. For companies that want to get a feel of what to expect, we therefore compiled a list of the most important topics addressed in the Commission’s proposal for the PPWR.
- Requirements for packaging to be recyclable. From 2030, all packaging will have to be ‘designed for recycling’ in accordance with state-of-the-art collection, sorting and recycling processes. As of 2035, packaging must be ‘recycled at scale’, meaning that packaging must be sufficiently and effectively collected, sorted and recycled in practice. Further details on the design for recycling and recycling at scale requirements shall follow by delegated acts adopted by the Commission.
- Minimum amount of recycled plastic content. From 2030, plastic packaging shall contain certain minimum amounts (depending on the type of packaging) of recycled plastic content. These amounts shall further increase by 2040. Instructions as to the methodology for the calculation and verification of the percentage of recycled content will follow by an implementing act. Where information on the recycled content is communicated on the packaging, harmonized labels shall be used for such.
- Mandatory industrial composability for certain packaging. Think of coffee and tea bags or other units, sticky labels attached to fruit and vegetables, and very lightweight plastic carrier bags. The list of packaging that need to be industrially compostable may be extended in future. Packaging that could have been designed as re-usable shall not be presented as compostable.
- Sorting instructions. Labels with information on the material composition shall be applied on packaging to help consumers identifying the appropriate disposal route.
- Rules on re-use and refill. Certain economic operators in the take-away and beverage sector will be subject to targets on re-use and refill. Re-usable packaging must fulfill a set of criteria, including being part of a system for re-use. Information facilitating re-use must be provided on the packaging via a QR code or otherwise. In case of products offered through refill, end-consumers shall be provided with information to ensure safe and hygienic use of the product.
- Increased rules for manufacturers to demonstrate compliance. Manufacturers manufacturing packaging under their own name or trademark, or having packaging designed or manufactured for use with their products, are subject to increased rules to demonstrate compliance with the PPWR. Where a manufacturer is supplied with packaging or packaging materials from a third party, such supplier must provide the manufacturer with all information necessary to demonstrate conformity.
- New roles of economic operators. A system of checks & balances is introduced by giving authorized representatives, importers and distributors specific verification tasks to ensure packaging is placed on the market in accordance with the PPWR.
- Harmonized criteria for modulated extended producer responsibility fees. Financial contributions to be paid by producers (those making available packaging for the first time in the EU under their own name of trademark) to take responsibility for the management of packaging at their end-of-life shall be modulated based on the recyclability of the packaging and the presence of recycled plastic content.
- Reduced empty space in e-commerce and other packaging. The weight and volume of packaging shall be minimized as much as possible. The ratio of empty space in e-commerce and other pre-defined packaging in relation to the packaged product(s) shall not exceed 40%.
- Prohibition on packaging in certain formats and for certain purposes. This mainly concerns certain single-use applications such as in the HORECA sector and for small amounts of fresh fruit and vegetables. The list of prohibited packaging is presented in Annex V to the proposed PPWR and can be amended by delegated act.
- A deposit-return system for single-use plastic beverage bottles and beverage cans. Where these systems do not yet exist for packaging up to 3 liters, such shall be implemented by 2029. For other packaging, Member States are encouraged to voluntarily set up deposit-return or other systems to enable re-use or refill.
Take-away for businesses
As follows from the above, EU food packaging legislation is currently under revision: sustainable packaging will be the new norm. Although the Communication is not and the proposal for the PPWR is not yet binding law, companies involved with food packaging are advised to take the published information serious and to prepare for the enactment of official legislation. This does not only mean staying up-to-date with upcoming legal requirements applicable to the business at stake, but also being ready to involve and control partners in the supply chain through (revised) legal contracts.
Posted: December 1, 2022 | Author: Jasmin Buijs | Filed under: Advertising, Enforcement, Food, Food Supplements, Health claims |
Intro
Food businesses operators that make medical claims for their products in the Netherlands can be fined for doing so under food law. However, they also run the risk of being fined under the Dutch Medicines Act (in Dutch: “Geneesmiddelenwet”), in which case much higher fine amounts apply. The latter sometimes provokes surprise and outrage. Based on three recent rulings, we see a positive trend, which is explained below.
Drug definition
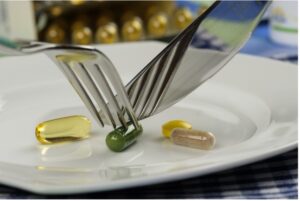 In principle, a medicinal product cannot be sold in the Netherlands without an authorization. Advertising a medicinal product that has not been authorized is prohibited as well. If a product is classified as such and sold without a license, the seller risks a hefty fine.
In principle, a medicinal product cannot be sold in the Netherlands without an authorization. Advertising a medicinal product that has not been authorized is prohibited as well. If a product is classified as such and sold without a license, the seller risks a hefty fine.
The legal definition of the term medicinal product and the corresponding authorization requirement can be found in the Dutch Medicines Act, which is based on the European Directive 2001/83/EC (the “Medicinal Product Directive”). The Medicinal Product Directive provides two criteria for the definition of a medicinal product: qualification by presentation and qualification by function. If a product meets one of these two criteria, it is classified as a medicinal product. The aforementioned criteria are further elaborated in case law.
Qualification by function
A product is a medicinal product by function (see the Hecht-Pharma judgment) if it can be administered to cure or prevent disease, diagnose or otherwise affect a person’s bodily functions. Of particular importance here are the composition and properties of the product, the method of use, the extent of distribution of the product, the consumers’ familiarity with the product and the health risks associated with its use.
Qualification by presentation
When applying the presentation criterion (see the Van Bennekom judgment), consideration is – unsurprisingly – given to whether a product should be regarded as a medicinal product on the basis of its presentation. It is not necessary that the product is expressly indicated or recommended as a medicinal product. The presentation criterion is already met if the manner of presentation gives the average consumer the impression that the product has a medicinal effect. The form in which the product is presented may give an indication for this, especially in the case of tablets, pills and capsules.
In particular the presentation criterion poses a risk to food companies. If they (unintentionally) make a medical claim in respect of their product, the presentation criterion may result in this product being classified (also) as a medicinal product by the Dutch Food and Consumer Product Safety Authority (“NVWA”). In that case, the NVWA may issue a fine under the Dutch Medicines Act. The starting point for such fine is €150,000, which is then differentiated based on the Policy rules of the Dutch Ministry of Health 2019. Even if the product also falls within the legal definition of food, the Dutch Medicines Act may apply simultaneously. The foregoing follows from the so-called hierarchy provision embodied in article 2.2 of the Medicinal Products Directive, which has been implemented into Dutch law as well. On the basis of this hierarchy principle, the Dutch Medicines Act is applicable if there is any doubt about the applicable product category. The result of this provision is that even a seller of coconut oil can receive a fine under the Dutch Medicines Act.
New trend in enforcement of medical claims?
Dutch case law gives numerous examples of products being classified as medicinal products by courts based on (solely) the presentation criterion. Recently, three court rulings have been rendered which give reason to assume that there is a new trend in case law. These are a ruling of the District Court of Oost-Brabant of March 25, 2022, and two (materially identical) rulings of the District Court of The Hague of June 28, 2022, regarding food supplements and follow-on milk, respectively.
The first case concerns the sale of dietary supplements, for which medical claims were made. The NVWA therefore classifies these supplements as medicinal products based on the presentation criterion and imposes two fines under the Dutch Medicines Act (both for sale and for advertising an unregistered medicinal product). The seller’s defense is that the Dutch Medicines Act should be interpreted in accordance with the Medicinal Product Directive and that it follows from there that the contested decision of the NVWA is based on an incorrect legal basis.
 The court agreed with this argumentation, referring to the amendment of the Medicinal Product Directive of 2004. The court deduces from the preamble to the amendment that the Medicinal Product Directive does not apply if there is no doubt that a product clearly exclusively belongs to another product category, such as food or food supplements. The court ruled that this was indeed the case for the specific circumstances that were under discussion. The products clearly fall under the category of food supplements and therefore solely food law applies. The court confirms that the Dutch Medicines Act must, after all, be interpreted in accordance with the Medicinal Product Directive. The court therefore does not proceed testing the medical claims made against the presentation criterion based on drug legislation at all.
The court agreed with this argumentation, referring to the amendment of the Medicinal Product Directive of 2004. The court deduces from the preamble to the amendment that the Medicinal Product Directive does not apply if there is no doubt that a product clearly exclusively belongs to another product category, such as food or food supplements. The court ruled that this was indeed the case for the specific circumstances that were under discussion. The products clearly fall under the category of food supplements and therefore solely food law applies. The court confirms that the Dutch Medicines Act must, after all, be interpreted in accordance with the Medicinal Product Directive. The court therefore does not proceed testing the medical claims made against the presentation criterion based on drug legislation at all.
Clearly food-only
The above ruling raises the question when a product is “clearly exclusively” a food and what aspects of the product are important in this respect. Indications for this can be found in the two recent decisions of the District Court of The Hague regarding specific food products for toddlers, namely follow-on formula. In its assessment of whether the follow-on formula in question could be a medicinal product by presentation, the court determined that such qualification is not obvious with regard to products sold in supermarkets and drugstores. Another factor in this case was that the detailed information about the follow-on formula, on the basis of which the Dutch Ministry of Health (the counterparty in the cases at stake) believed it to be a medicinal product by presentation, could only be found on the seller’s website.
Conclusion
Based on the rulings discussed, we signal a trend that judges are halting the current practice of enforcement of prohibited medical claims for food products based on the Dutch Medicines Act. The discussed rulings make clear that (prohibited) health claims for food supplements and for other food products such as follow-on formula should be assessed on the basis of the Food Information for Consumers Regulation (the “FIC Regulation”), and not via the presentation criterion based on the Dutch Medicines Act. In our opinion this is justified, because since the FIC Regulation became applicable, food law is specifically set up to do so. We are very curious to see whether the trend initiated above will be followed by other courts. Although it follows from a ruling of the District Court of Zeeland-West-Brabant of 21 October, 2022, that this is not yet the case, we trust this will only be a matter of time.
The above does however not mean that food business operators would be allowed to make medical claims for their products. Also, the FIC Regulation contains a ban on medical claims for food products and the Nutrition and Health Claims Regulation sets forth a strict regime for authorized health claims. Having said that, fines following a violation of food legislation are far lower than fines based on the Dutch Medicines Act. On balance, food companies are therefore better off with fines based on food legislation.
This blogpost is written by Max Baltussen, Karin Verzijden and Jasmin Buijs.
The authors want to acknowledge Ebba Hoogenraad and Irene Verheijen for sharing the case law discussed here.
 Claims for formula milk that refer to nature can be understood as a (prohibited) discouragement of breastfeeding, as was recently ruled in two instances by the Dutch Advertising Code Foundation. This case captured our attention, especially with an eye to future possibilities of cell-based breastmilk alternatives.
Claims for formula milk that refer to nature can be understood as a (prohibited) discouragement of breastfeeding, as was recently ruled in two instances by the Dutch Advertising Code Foundation. This case captured our attention, especially with an eye to future possibilities of cell-based breastmilk alternatives. Our analysis and future outlook: It is commonly agreed that breastfeeding, where possible, should be supported. This is one of the principles laid down in the WHO International Code of Marketing of Breast-Milk Substitutes and the subsequent relevant resolutions of the World Health Assembly. We are therefore not surprised by the ruling.
Our analysis and future outlook: It is commonly agreed that breastfeeding, where possible, should be supported. This is one of the principles laid down in the WHO International Code of Marketing of Breast-Milk Substitutes and the subsequent relevant resolutions of the World Health Assembly. We are therefore not surprised by the ruling. Last month, the conference Regulating the Future of Foods took place in Barcelona, gathered more than hundred professionals active in the fields of precision fermentation and cellular agriculture. The purpose of the conference was to define hurdles and investigate opportunities in the current regulatory framework applicable to this sector. Many interesting presentations took place discussing the global perspective of our future food system and AXON moderated a workshop targeting tastings of cultivated foods, formulating a number of conversation topics. In this blogpost, we share the outcome of the discussions that took place during this workshop.
Last month, the conference Regulating the Future of Foods took place in Barcelona, gathered more than hundred professionals active in the fields of precision fermentation and cellular agriculture. The purpose of the conference was to define hurdles and investigate opportunities in the current regulatory framework applicable to this sector. Many interesting presentations took place discussing the global perspective of our future food system and AXON moderated a workshop targeting tastings of cultivated foods, formulating a number of conversation topics. In this blogpost, we share the outcome of the discussions that took place during this workshop. Germany’s highest court, the Bundesgerichtshof, asked the European Court of Justice (ECJ) this summer to explain the use of ‘on hold’ claims for so-called ‘botanicals’. The question is whether these substances may be advertised with health claims or general, non-specific health benefits as long as the assessment by EFSA has not been completed and the European Commission has not yet taken a final decision on the authorization of these claims.
Germany’s highest court, the Bundesgerichtshof, asked the European Court of Justice (ECJ) this summer to explain the use of ‘on hold’ claims for so-called ‘botanicals’. The question is whether these substances may be advertised with health claims or general, non-specific health benefits as long as the assessment by EFSA has not been completed and the European Commission has not yet taken a final decision on the authorization of these claims. Whether the aforementioned alternative views are sufficient to exclude botanicals from the scope of Article 10(1) and (3) Claims Regulation remains to be seen. As highlighted in a report published last September, companies are able to make health claims for botanicals that are included in the ‘on hold’ list under the transitional regime of Article 28(5) and (6) of the Claims Regulation. Moreover, the ECJ ruled in previous cases that companies making ‘on hold’ claims are not disproportionately disadvantaged. Since they can make claims without EFSA having assessed these and/or without a final decision from the European Commission, they are in fact favored.
Whether the aforementioned alternative views are sufficient to exclude botanicals from the scope of Article 10(1) and (3) Claims Regulation remains to be seen. As highlighted in a report published last September, companies are able to make health claims for botanicals that are included in the ‘on hold’ list under the transitional regime of Article 28(5) and (6) of the Claims Regulation. Moreover, the ECJ ruled in previous cases that companies making ‘on hold’ claims are not disproportionately disadvantaged. Since they can make claims without EFSA having assessed these and/or without a final decision from the European Commission, they are in fact favored. In principle, a medicinal product cannot be sold in the Netherlands without an authorization. Advertising a medicinal product that has not been authorized is prohibited as well. If a product is classified as such and sold without a license, the seller risks a hefty fine.
In principle, a medicinal product cannot be sold in the Netherlands without an authorization. Advertising a medicinal product that has not been authorized is prohibited as well. If a product is classified as such and sold without a license, the seller risks a hefty fine. The court agreed with this argumentation, referring to the amendment of the Medicinal Product Directive of 2004. The court deduces from the preamble to the amendment that the Medicinal Product Directive does not apply if there is no doubt that a product clearly exclusively belongs to another product category, such as food or food supplements. The court ruled that this was indeed the case for the specific circumstances that were under discussion. The products clearly fall under the category of food supplements and therefore solely food law applies. The court confirms that the Dutch Medicines Act must, after all, be interpreted in accordance with the Medicinal Product Directive. The court therefore does not proceed testing the medical claims made against the presentation criterion based on drug legislation at all.
The court agreed with this argumentation, referring to the amendment of the Medicinal Product Directive of 2004. The court deduces from the preamble to the amendment that the Medicinal Product Directive does not apply if there is no doubt that a product clearly exclusively belongs to another product category, such as food or food supplements. The court ruled that this was indeed the case for the specific circumstances that were under discussion. The products clearly fall under the category of food supplements and therefore solely food law applies. The court confirms that the Dutch Medicines Act must, after all, be interpreted in accordance with the Medicinal Product Directive. The court therefore does not proceed testing the medical claims made against the presentation criterion based on drug legislation at all.












 The WFD (currently under
The WFD (currently under